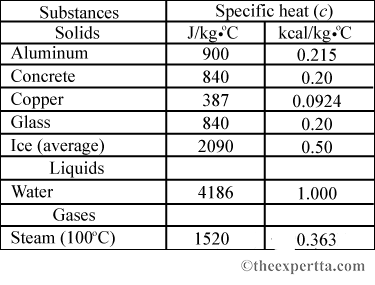
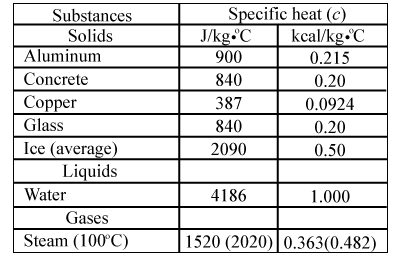
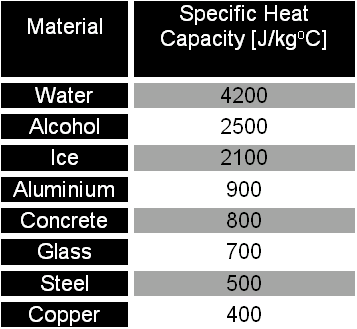
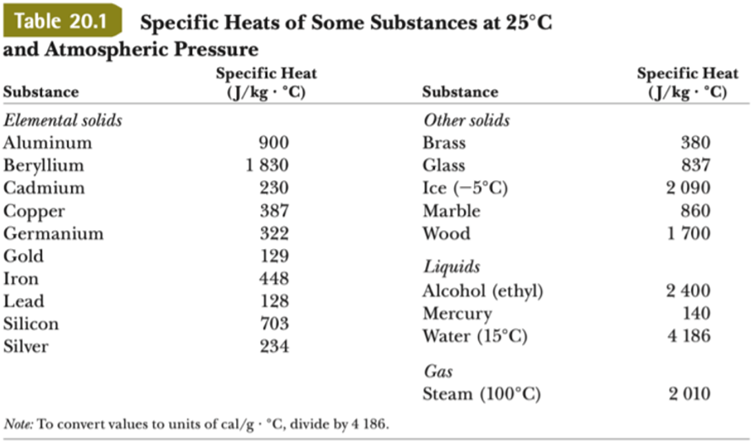
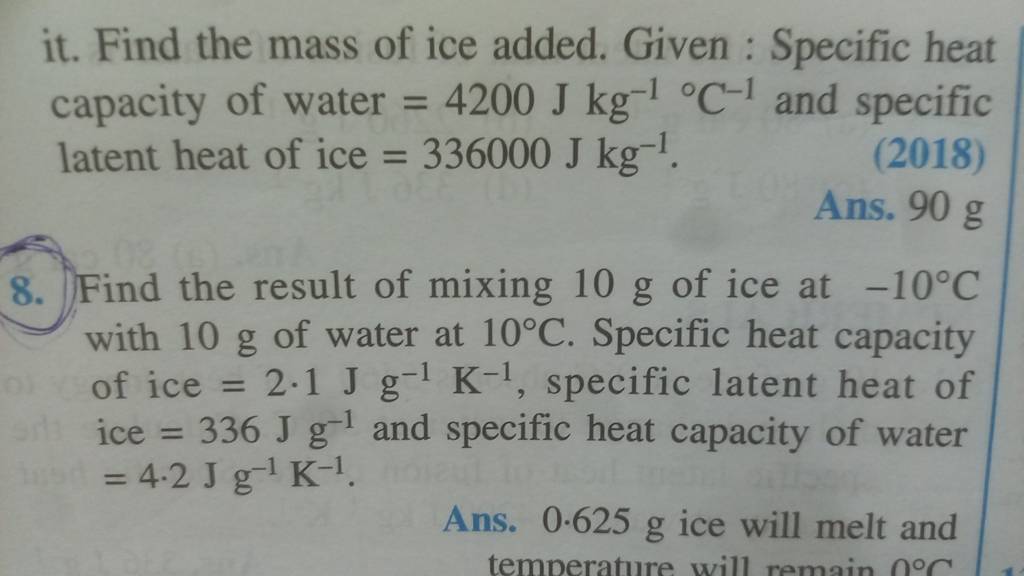Specific heat capacity Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online

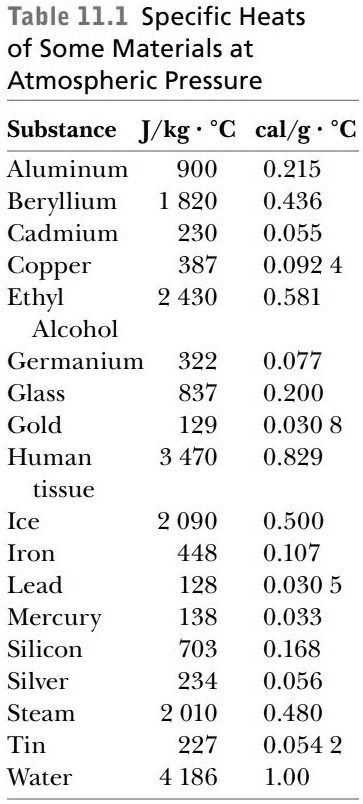
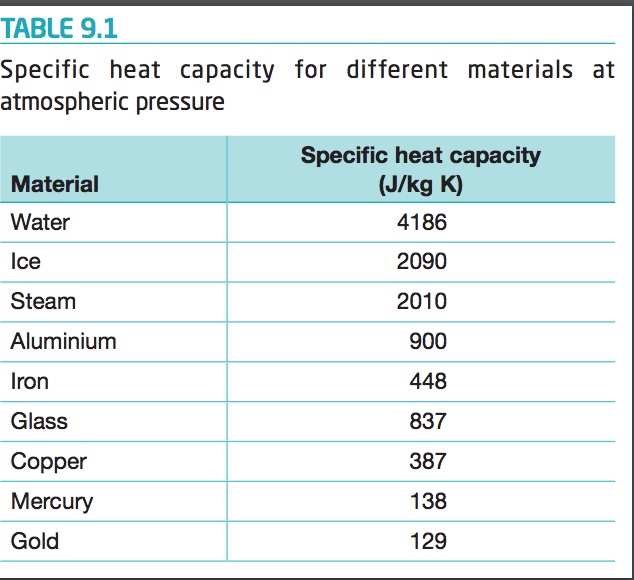
SOLVED: Specific heat capacity of water = 4186 J kg^-1 K^-1 Specific heat capacity of ice = 2090 J kg^-1 K^-1 Latent heat of fusion of ice = 335 kJ kg^-1 Latent

Calculate the heat required to convert 3 kg of ice at `-12^()C` kept in a calorimeter to steam a... - YouTube

The temperature of 1.94 kg of water is 34 °C. To cool the water, ice at 0°C is added to it. The desired - brainly.com
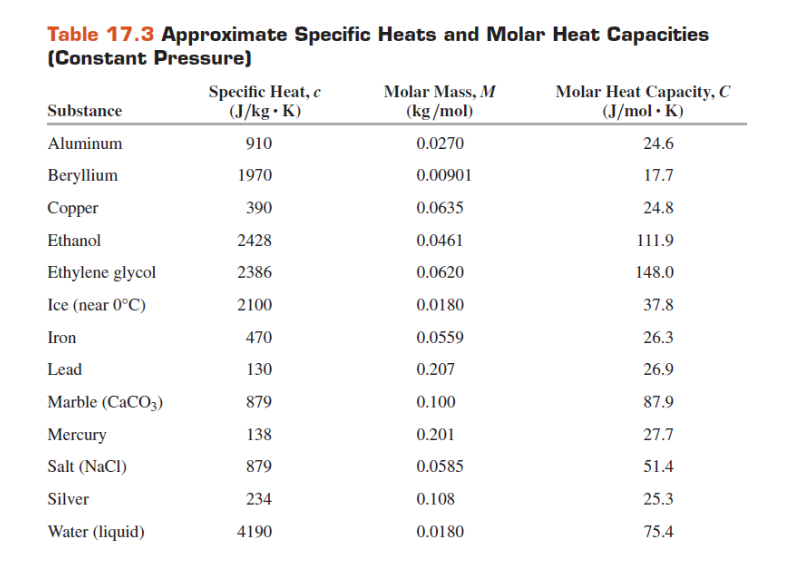
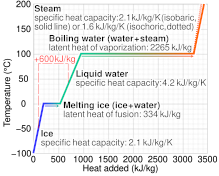
What are the values of the specific heat capacity of water, steam, and ice (in both joule and cal.) and the values of latent heat of fusion and vaporisation for water? -

A 40 g ice cube (latent heat of fusion = 3.33 x 10^5 J/kg) floats in 200 g of water (specific heat = 4184 J/kg degree Celsius) in a 100 g copper (

When you heat a substance, you are transferring energy into it by placing it in contact with surroundings that have a higher temperature. - ppt download

Calculate the heat required to convert 3 kg of ice at - 12^o C kept in a calorimeter to steam at 100^o at atmospheric pressure. (Given: specific heat of ice = 2.100 ×

The amount of heat energy required to convert 1 kg of ice at - 10^∘C to water at 100^∘C is 7,77,000 J. Calculate the specific latent heat of ice. Specific heat capacity

The amount of heat energy required to convert 1 kg of ice at - 10^∘C to water at 100^∘C is 7,77,000 J. Calculate the specific latent heat of ice. Specific heat capacity

1 PHYS1001 Physics 1 REGULAR Module 2 Thermal Physics HEAT CAPACITY LATENT HEAT What is cooking all about? ptC_heat.ppt. - ppt download